高通量單細(xì)胞水平細(xì)胞間相互作用自動(dòng)分析系統(tǒng) 該平臺提供了對單個(gè)細(xì)胞如何移動(dòng)、行為、交互和表現(xiàn)的高通量評估 
了解哪些細(xì)胞表現(xiàn)佳以及原因 評估細(xì)胞激活、殺傷和運(yùn)動(dòng)間的變化 一可同時(shí)評估單個(gè)細(xì)胞如何移動(dòng)、激活、相互作用、殺死和生存的單細(xì)胞技術(shù) 科學(xué)家依靠該系統(tǒng)平臺量化數(shù)千個(gè)免疫細(xì)胞的時(shí)空性能,以了解哪些候選細(xì)胞進(jìn)入臨床試驗(yàn)、了解臨床反應(yīng)并保持生產(chǎn)的一致性
提供單細(xì)胞的面、動(dòng)態(tài)分析。研究細(xì)胞活動(dòng)的其他方法缺乏將動(dòng)態(tài)細(xì)胞行為與單細(xì)胞水平的分子行為整合的能力。該納米孔網(wǎng)格延時(shí)成像顯微鏡平臺應(yīng)用視覺 AI 來評估細(xì)胞激活、殺傷和運(yùn)動(dòng)作為時(shí)間的函數(shù),以便大限度地了解細(xì)胞功能、狀態(tài)和表型。
平臺廣泛適用于各種細(xì)胞類型和應(yīng)用。雖然生命科學(xué)研究人員受益于單細(xì)胞分析的重大,但該系統(tǒng)超越了靜態(tài)分析。通過該系統(tǒng),生命科學(xué)創(chuàng)新者可以觀察新的生物學(xué),并對細(xì)胞如何移動(dòng)、激活、殺死和生存獲得新的、意想不到的理解。
納米阱網(wǎng)格是一種廣泛適用的、實(shí)用的方法,用于記錄體外動(dòng)態(tài)細(xì)胞-細(xì)胞相互作用,提供了廣泛統(tǒng)計(jì)采樣所需的大量吞吐量。重要的是,當(dāng)圖像分析揭示出特別感興趣的細(xì)胞時(shí),它們的坐標(biāo)是足夠的,可以使機(jī)器人檢索用于克隆擴(kuò)增或PCR等下游處理。在癌癥免疫治療的背景下,該方法可以監(jiān)測效應(yīng)器介導(dǎo)的對所需靶細(xì)胞的細(xì)胞毒性,而無需靶細(xì)胞工程。這提供了重要的優(yōu)勢,例如使用自體或匹配/原代腫瘤細(xì)胞作為靶細(xì)胞的能力。 該系統(tǒng)從納米阱網(wǎng)格中細(xì)胞的高通量延時(shí)成像顯微鏡數(shù)據(jù)中自動(dòng)分析細(xì)胞-細(xì)胞相互作用,為免疫療法中的細(xì)胞-細(xì)胞交互作用提供了基本見解。
該系統(tǒng)廣泛適用于一般免疫學(xué)、細(xì)胞生物制造、癌癥生物學(xué)、分化、干細(xì)胞工程、基于血液中不同細(xì)胞類型之間相互作用的藥物篩選中動(dòng)態(tài)細(xì)胞-細(xì)胞相互作用的高通量定量研究。 該系統(tǒng)執(zhí)行大規(guī)模自動(dòng)化視頻陣列分析。它提供了一個(gè)快速的“交鑰匙”解決方案,具有高效的用戶界面,以單細(xì)胞分辨率生成細(xì)胞相互作用行為的定量分析, 具有高產(chǎn)量、自動(dòng)化、速度和準(zhǔn)確性,并具有高效的視覺確認(rèn)。 它使用了利用納米阱空間限制的分割和跟蹤算法,實(shí)現(xiàn)了98%以上的細(xì)胞檢測、分割和跟蹤精度,而這些精度是無法直接實(shí)現(xiàn)的。該系統(tǒng)在集中式服務(wù)器上運(yùn)行, 需要在服務(wù)器之間進(jìn)行耗時(shí)的數(shù)據(jù)傳輸。 它自動(dòng)計(jì)數(shù)每個(gè)納米井中的細(xì)胞,根據(jù)細(xì)胞類型標(biāo)記識別細(xì)胞類型,并提供細(xì)胞大小、位置、形狀和運(yùn)動(dòng)的動(dòng)態(tài)測量;細(xì)胞-細(xì)胞接觸的頻率和持續(xù)時(shí)間, 以及細(xì)胞事件熒光標(biāo)記物的變化。它實(shí)現(xiàn)了這些測量的鏈接可視化和分析,并具有快速編輯功能。為了實(shí)現(xiàn)下游統(tǒng)計(jì)分析和假設(shè)測試,我們軟件中的數(shù)據(jù)可以以電子表格的形式導(dǎo)出 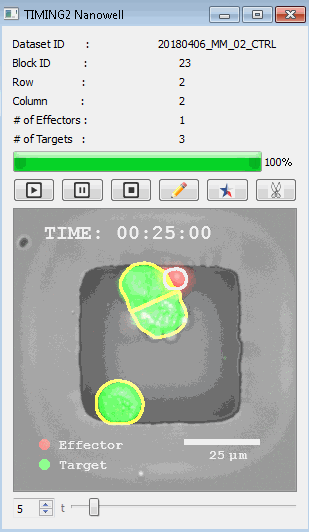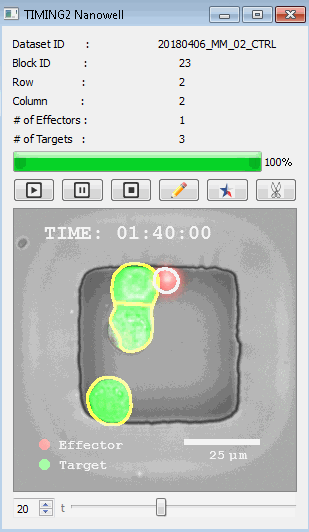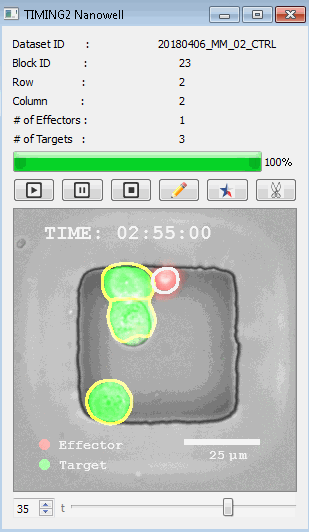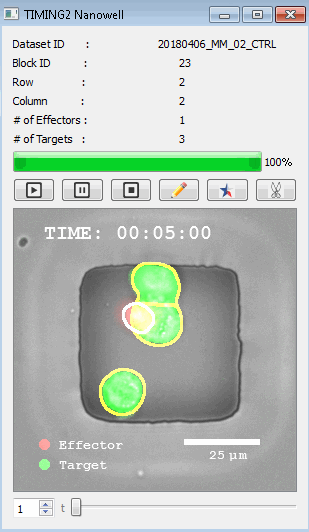 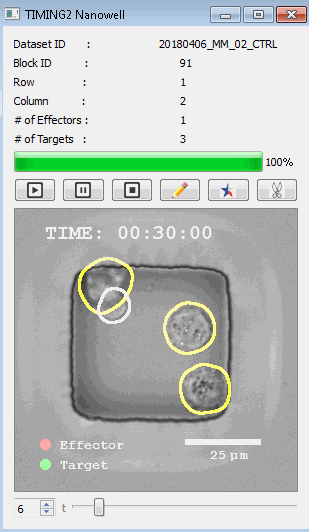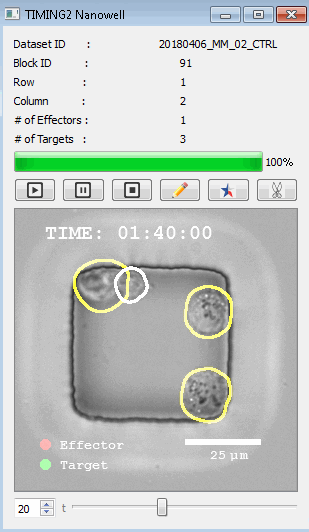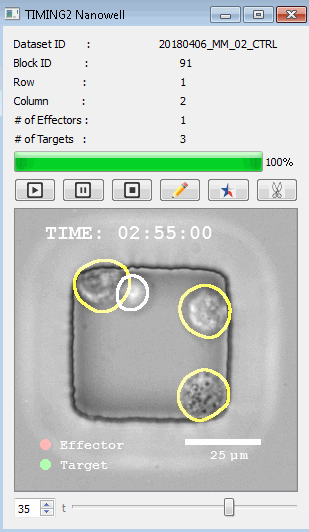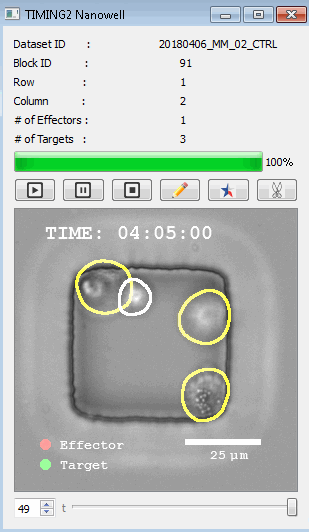 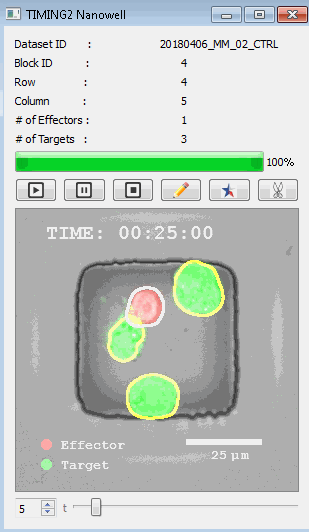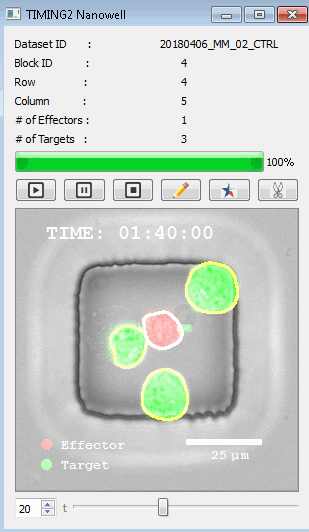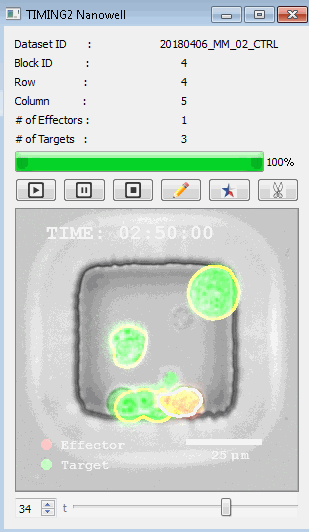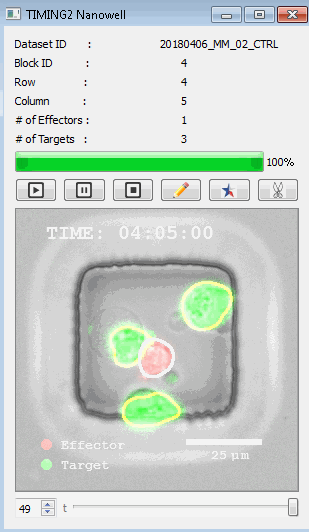 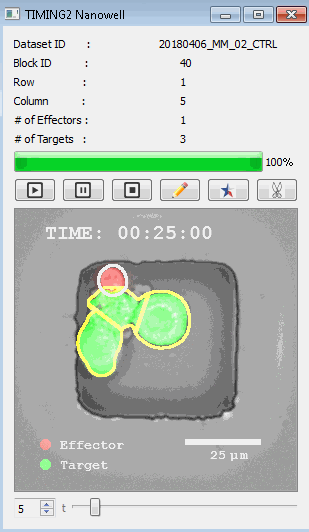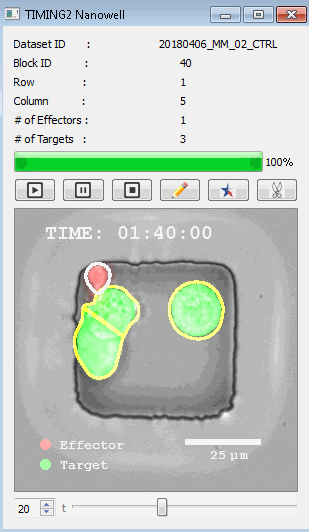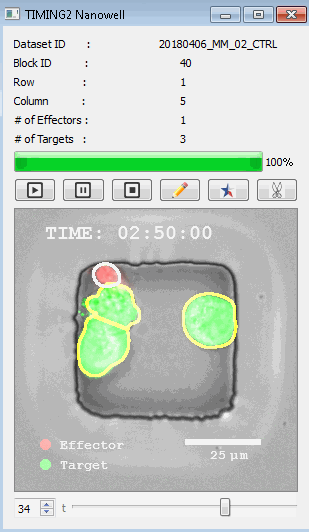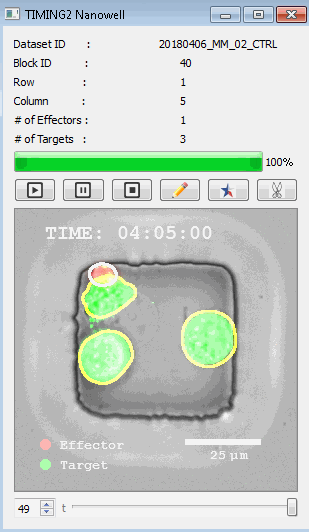 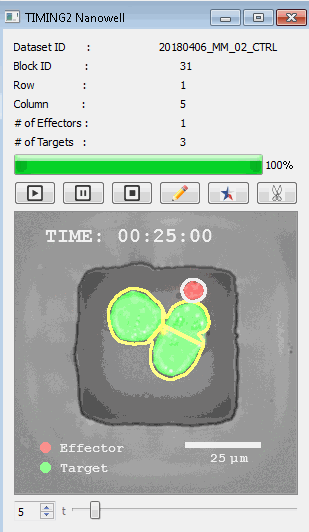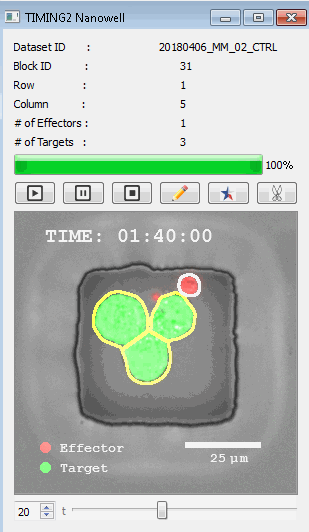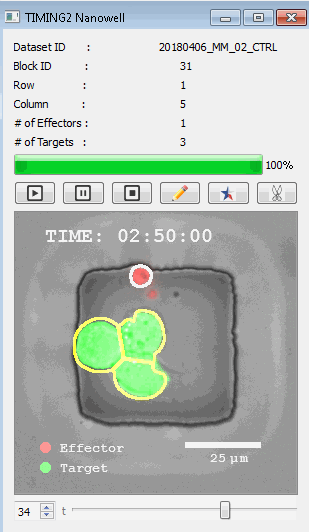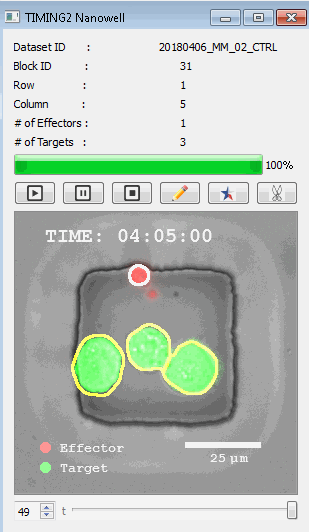 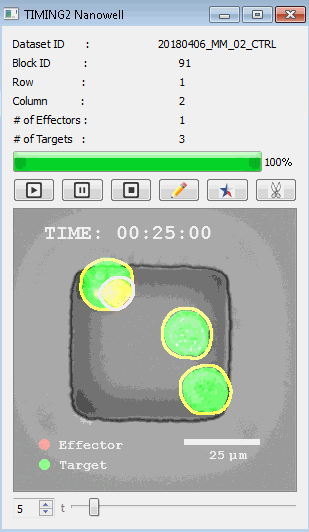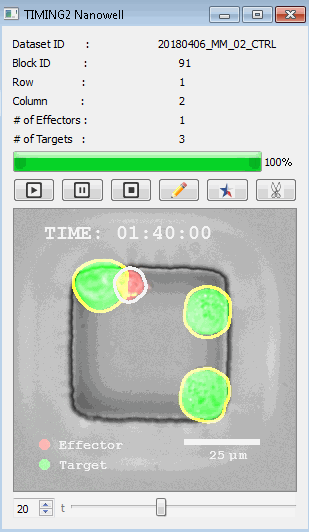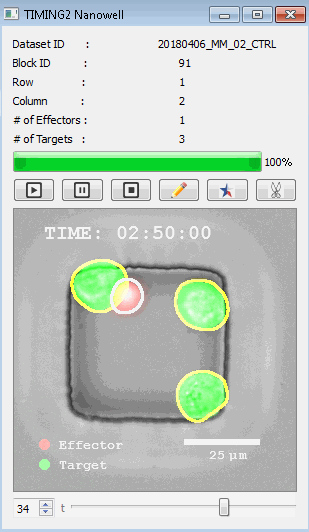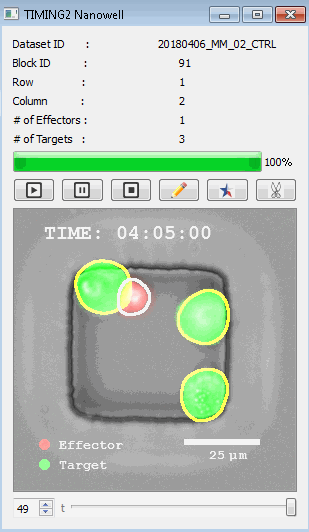
工作流程: 從客戶處收到冷凍保存的效應(yīng)細(xì)胞和靶細(xì)胞后,該系統(tǒng)解凍并進(jìn)行細(xì)胞系活力研究。還可以運(yùn)行客戶特定的檢測,以確保細(xì)胞以預(yù)期的方式表現(xiàn)。此時(shí),執(zhí)行其單細(xì)胞工作流程如下:
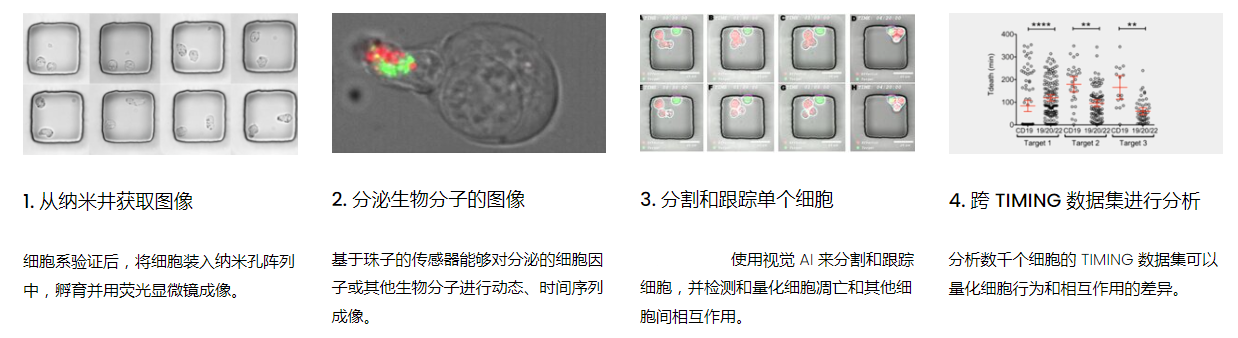
DISCover 工作流程 TIMING分析使該系統(tǒng)能夠識別感興趣的細(xì)胞以進(jìn)行進(jìn)一步分析,從而更面地了解細(xì)胞的行為方式。
直觀地評估細(xì)胞間的表現(xiàn)和相互作用的情況 納米孔網(wǎng)格延時(shí)成像顯微鏡 (TIMING) 應(yīng)用基于神經(jīng)網(wǎng)絡(luò)的檢測來評估數(shù)千個(gè)單個(gè)細(xì)胞與細(xì)胞的相互作用
表征數(shù)千個(gè)細(xì)胞的遷移、接觸動(dòng)力學(xué)和細(xì)胞死亡
該系統(tǒng)應(yīng)用人工智能并行進(jìn)行數(shù)千個(gè)顯微鏡實(shí)驗(yàn),以高通量和單細(xì)胞分辨率表征遷移、細(xì)胞間相互作用、細(xì)胞毒性、存活和生物分子分泌 
將運(yùn)動(dòng)和性能與多組學(xué)分析相結(jié)合 可以識別和檢索感興趣的細(xì)胞,將功能分析與單細(xì)胞 RNA 測序和流式細(xì)胞術(shù)等分析方式的信息聯(lián)系起來,從而提供對細(xì)胞功能、狀態(tài)和表型的面了解。 
The CellChorus platform is the only approach that can associate motility, contact, killing, cytokine secretion and other dynamic readouts over time for individual cells and cell-cell interactions at high throughput. This approach is called dynamic single-cell analysis. The platform has broad application across antibodies, cell therapies, vaccines and other technologies, as shown in the publications below from leading journals such as Blood, Clinical Cancer Research, Nature Immunology, the JITC, Journal of Immunology, and Science Advances. In less than 18 months, the FuseBio team developed a TCE lead candidate that targets ROR1 (FUSE394). The data presented at SITC 2022 demonstrated that FUSE394 is potent and successfully decouples anti-tumor activity from cytokine release and T cell exhaustion. TIMING data from the CellChorus early access program demonstrated that FUSE394 preserves T cell motility and healthy morphology and promotes 50% more synapse formation than a non-decoupled control. Researchers applied the TIMING platform to test NK-mediated cytotoxicity at single-cell resolution as part of research showing that chimeric antigen receptor (CAR) activation in natural killer (NK) cells promoted transfer of the CAR cognate antigen from tumor to NK cells, resulting in (1) lower tumor antigen density, thus impairing the ability of CAR-NK cells to engage with their target, and (2) induced self-recognon and continuous CAR-mediated engagement, resulting in fratricide of trogocytic antigen-expressing NK cells (NKTROG+) and NK cell hyporesponsiveness. Researchers applied the CellChorus TIMING platform to evaluate interactions between thousands of tumor cells and CAR-T cells that were to be infused into patients to preview how the CAR-T cells being given to patients can eradicate tumor cells after infusion. The researchers robotically selected super-killer T cells for transcriptional profiling to characterize the molecular properties of the CAR-T cells with the best features for eradicating tumors. Time-lapse imaging microscopy in nanowell grids (TIMING?) profiling revealed that T cells from responders showed migration (persistent motion for at least one body length), and migration was associated with serial killing capacity. In addon, confocal microscopy revealed that migration is linearly correlated with both mitochondrial volume and lysosomal volume; and scRNA-seq demonstrated that T cells from responders were enriched in pathways related to T-cell killing, migration and actin cytoleton, and TCR clustering. By simultaneously evaluating thousands of individual interactions between T cells and target cells bearing virally derived peptides, the company’s artificial intelligence-powered TIMING? platform reveals individual T cells that are capable of polyfunctionality based on killing, serial killing, and secretion of the cytokine interferon gamma (IFNγ). Time-lapse imaging in nanoliter wells was applied to understand the kinetics of translocation at the single‐cell level and to enable tracking of the same individual cells. The micromesh array contains nanoliter wells that enabled imaging protein translocation dynamically. Understanding why only some T cells are capable of killing, and identifying mechanisms that can improve killing has remained elusive. These results illustrate that while non-killer T cells are reflective of population heterogeneity, integrated single-cell profiling can enable identification of mechanisms that can enhance the function/proliferation of killer T cells leading to direct anti-tumor benefit. Using time-lapse imaging microscopy to monitor T cell-mediated tumor killing at the single cell level to gain more complete understanding of the kinetics of killing in studies that reveal two distinct types of immune resistance regulators and demonstrate their potential as therapeutic targets to improve the efficacy of immunotherapy. Immune cells such as T cells and natural killer cells, and target cells such as NALM6, K562 and EL4, were incubated in PDMS nanowell arrays and imaged using time-lapse fluorescent microscopy. The proposed cell segmentation and tracking algorithms allowed automated quantification of cell pairs, cell location, morphology, interactions and movement as well as cell viability, without the need for manual processing. The result revealed that cytotoxic T cells have higher motility than noncytotoxic T cells, both before and during synapse formation. We found sustained activation of cytotoxicity, costimulation, oxidative phosphorylation– and proliferation-related genes, and simultaneously reduced differentiation and exhaustion. Our study identifies molecular features of TCR8 expression that can guide the development of enhanced immunotherapies. CD19/20/22CAR T-cells killed CD19(?) blasts from patients who relapsed after CD19CAR T-cell therapy and CRISPR/Cas9 CD19 knockout primary BL-ALL both in vitro and in an animal model, while CD19CAR T-cells were ineffective. At the subcellular level, CD19/20/22CAR T-cells formed dense immune synapses with target cells that mediated effective cytolytic complex formation, were efficient serial killers in single-cell tracking studies, and were as efficacious as CD19CAR T-cells against primary CD19(+) disease. In conclusion, independent of CD19 expression, CD19/20/22CAR T-cells could be used as salvage or front-line CAR therapy for patients with recalcitrant disease. By utilizing both tradonal blob detection to generate binary mask labels from the stained channel images and the deep learning Mask RCNN model to train a detection and segmentation model, we managed to segment nuclei based only on phase images. The detection average precision is 0.82 when the IoU threshold is to be set 0.5. And the mean IoU for masks generated from phase images and ground truth masks from experts is 0.735. Without any ground truth mask labels during the training time, this is good enough to prove our hypothesis. This result enables the ability to detect nuclei without the need for exogenous labeling. This paper proposes an efficient variant of capsule networks (CapsNets) as an alternative to CNNs. Extensive experimental results demonstrate that the proposed CapsNets achieve competve performances in target cell apoptosis classification, while significantly outperforming CNNs when the number of training samples is small. To utilize temporal information within microscopy videos, we propose a recurrent CapsNet constructed by stacking a CapsNet and a bi-directional long short-term recurrent structure. Our experiments show that when considering temporal constraints, the recurrent CapsNet achieves 93.8% accuracy and makes significantly more consistent prediction than NNs. Integration of transcriptomic profiling, immune phenotyping and metabolism demonstrated that motile cells are more na?ve-like with higher oxidative metabolism and spare respiratory capacity. Our result also revealed that the master metabolic regulator AMP kinase (AMPK) is required for CAR+ T cells with high motility. We used a xenograft leukemia mouse model (CD19+ NALM-6) and validated that the motile cells have enhanced persistence and superior anti-cancer effect in vivo compared to the parental un-sorted population. Collectively, our multi-dimensional results demonstrated that persistent motility is a selectable biomarker of expanded CAR+ T cell bioactivity. On a desktop computer, TIMING 2.0 takes 5 s/block/image frame, four times faster than our previous method on the same computer, and twice as fast as our previous method (TIMING) running on a Dell PowerEdge server. The cell segmentation accuracy (f-number?=?0.993) is superior to our previous method (f-number?=?0.821). A graphical user interface provides the ability to inspect the video analysis results, make corrective edits efficiently (one-click edng of an entire nanowell video sequence in 5–10 s) and display a summary of the cell killing efficacy measurements. Genetically engineered T cells that express chimeric antigen receptors (CAR+) are heterogeneous and thus, understanding the immunotherapeutic efficacy remains a challenge in adoptive cell therapy. We developed a high-throughput single-cell methodology, Timelapse Imaging Microscopy In Nanowell Grids (TIMING) to monitor interactions between immune cells and tumor cells in vitro. Using TIMING we demonstrated that CD4+ CAR+ T cells participate in multi-killing and benefit from improved resistance to activation induced cell death in comparison to CD8+ CAR+ T cells. For both subsets of cells, effector cell fate at the single-cell level was dependent on functional activation through multiple tumor cells. A comprehensive understanding of the polyfunctionality of T lymphocytes in ICI or adoptive cell transfer (ACT), at single-cell resolution, will quantify T-cell properties that are essential for therapeutic benefit. We briefly highlight several emerging integrated single-cell technologies focusing on the profiling of multiple properties/functionales of T cells. We envision that these tools have the potential to provide valuable experimental and clinical insights on T-cell biology, and eventually pave the road for the discovery of surrogate T-cell biomarkers for immunotherapy. CD8+BTLA- TILs could not control tumor growth in vivo as well as their BTLA+ counterpart and antigen-specific CD8+BTLA- T cells had impaired recall response to a vaccine. However, CD8+BTLA+ TILs displayed improved survival following the killing of a tumor target and heightened "serial killing" capacity. Using mutants of BTLA signaling motifs, we uncovered a costimulatory function mediated by Grb2 through enhancing the secretion of IL-2 and the activation of Src after TCR stimulation. Our data portrays BTLA as a molecule with the singular ability to provide both costimulatory and coinhibitory signals to activated CD8+ T cells, resulting in extended survival, improved tumor control, and the development of a functional recall response. Clin Cancer Res; 23(20); 6151-64. ?2017 AACR. We used the engineered Fc domains to demonstrate in vitro and in mouse models that for therapeutic antibodies, complement-dependent cell-mediated cytotoxicity (CDCC) and complement-dependent cell-mediated phagocytosis (CDCP) by immunological effector molecules mediated the clearance of target cells with kinetics and efficacy comparable to those of the FcγR-dependent effector functions that are much better studied, while they circumvented certain adverse reactions associated with FcγR engagement. Collectively, our data highlight the importance of CDCC and CDCP in monoclonal-antibody function and provide an experimental approach for delineating the effect of complement-dependent effector-cell engagement in various therapeutic settings. Analysis of hundreds of individual human peripheral blood NK cells profiled ex vivo revealed that CD56dimCD16+ NK cells are immediate secretors of interferon gamma (IFN-γ) upon activation by phorbol 12-myristate 13-acetate (PMA) and ionomycin (< 3 h), and that there was no evidence of cooperation between NK cells leading to either synergistic activation or faster IFN-γ secretion. Furthermore, we observed that both the amount and rate of IFN-γ secretion from individual NK cells were donor-dependent. Collectively, these results establish our methodology as an investigational tool for combining phenotyping and real-time protein secretion of individual cells in a high-throughput manner. In aggregate, these results demonstrate the utility of our TIMING single cell methodology in uncovering not only the dynamic profile of T-cell behavior but also the ability to identify subpopulations of T-cell with enhanced polyfunctionality. Our studies support the use of motility as a surrogate and selective marker of higher CAR+ T cell bioactivity. These results also open up avenues to molecularly engineer T cells for an increased motility that could translate to better in vivo outcomes. Fluorescently labeled human T cells, natural killer cells (NK), and various target cells (NALM6, K562, EL4) were co-incubated on polydimethylsiloxane arrays of sub-nanoliter wells (nanowells), and imaged using multi-channel time-lapse microscopy. The proposed cell segmentation and tracking algorithms account for cell variability and exploit the nanowell confinement property to increase the yield of correctly analyzed nanowells from 45% (existing algorithms) to 98% for wells containing one effector and a single target, enabling automated quantification of cell locations, morphologies, movements, interactions, and deaths without the need for manual proofreading. Automated analysis of recordings from 12 different experiments demonstrated automated nanowell delineation accuracy >99%, automated cell segmentation accuracy >95%, and automated cell tracking accuracy of 90%, with default parameters, despite variations in illumination, staining, imaging noise, cell morphology, and cell clustering. An example analysis revealed that NK cells efficiently discriminate between live and dead targets by altering the duration of conjugation. The data also demonstrated that cytotoxic cells display higher motility than non-killers, both before and during contact. We implemented Timelapse Imaging Microscopy in Nanowell Grids (TIMING) to provide direct evidence that CD4(+)CAR(+) T cells (CAR4 cells) can engage in multikilling via simultaneous conjugation to multiple tumor cells. Comparisons of the CAR4 cells and CD8(+)CAR(+) T cells (CAR8 cells) demonstrate that, although CAR4 cells can participate in killing and multikilling, they do so at slower rates, likely due to the lower granzyme B content. Significantly, in both sets of T cells, a minor subpopulation of individual T cells identified by their high motility demonstrated efficient killing of single tumor cells. A comparison of the multikiller and single-killer CAR(+) T cells revealed that the propensity and kinetics of T-cell apoptosis were modulated by the number of functional conjugations. T cells underwent rapid apoptosis, and at higher frequencies, when conjugated to single tumor cells in isolation, and this effect was more pronounced on CAR8 cells. Our results suggest that the ability of CAR(+) T cells to participate in multikilling should be evaluated in the context of their ability to resist activation-induced cell death. We anticipate that TIMING may be used to rapidly determine the potency of T-cell populations and may facilitate the design and manufacture of next-generation CAR(+) T cells with improved efficacy. We demonstrate that the DLE-HuM195 antibody increases both the quality and the quantity of NK cell-mediated antibody-dependent cytotoxicity by endowing more NK cells to participate in cytotoxicity via accrued CD16-mediated signaling and by increasing serial killing of target cells. NK cells encountering targets coated with DLE-HuM195 induce rapid target cell apoptosis by promoting simultaneous conjugates to multiple target cells and induce apoptosis in twice the number of target cells within the same period as the wild-type mAb. Enhanced target killing was also associated with increased frequency of NK cells undergoing apoptosis, but this effect was donor-dependent. Antibody-based therapies targeting tumor antigens will benefit from a better understanding of cell-mediated tumor elimination, and our work opens further opportunes for the therapeutic targeting of CD33 in the treatment of acute myeloid leukemia.
|

































